Description
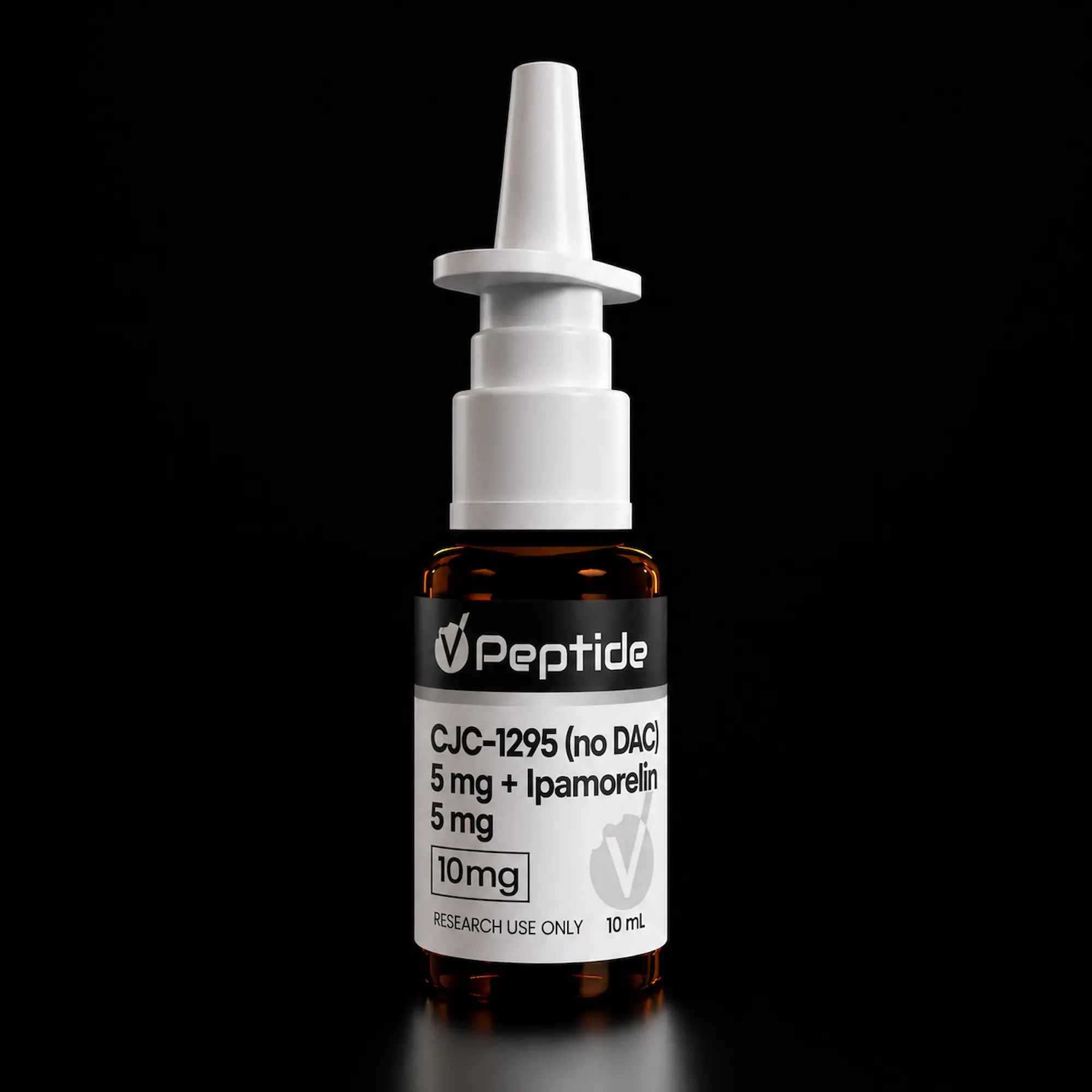
This research blend combines two complementary growth hormone–axis peptides in a single nasal spray formulation: CJC-1295 (no DAC), a modified GHRH(1-29) analog, and ipamorelin, a selective ghrelin/GHS-R agonist. The pairing allows concurrent investigation of GHRH receptor and growth hormone secretagogue receptor pathways from two structurally distinct mechanisms. CJC-1295 (no DAC), also known as Mod GRF 1-29, is a 29-amino-acid GHRH analog with four substitutions (D-Ala², Gln⁸, Ala¹⁵, Leu²⁷) designed to resist DPP-IV cleavage while maintaining a short pulsatile half-life. Ipamorelin is a synthetic pentapeptide studied for its selective ghrelin receptor activation with minimal off-target activity at cortisol, prolactin, ACTH, and aldosterone pathways.
Product Highlights
• Each vial contains 10 mL solution (~100 sprays)
• Each spray delivers 0.1 mL = 50 mcg CJC-1295 (no DAC) + 50 mcg Ipamorelin
• Total CJC-1295 (no DAC) Content: 5 mg per vial
• Total Ipamorelin Content: 5 mg per vial
• Total Peptide Content: 10 mg per vial
• Specialized pharmaceutical-grade nasal solution — designed for comfort and ease on the nose
• Unlike many other nasal sprays, this formula avoids irritation and dryness
• Packaged in Class A, lead-free pharmaceutical glass bottles
• Tamper/Child-Proof Cap + Orifice Reducer for integrity, sterility, and ease of use
Description
This dual-peptide nasal spray is explored for combined activation of the GHRH receptor (via CJC-1295 no DAC) and the growth hormone secretagogue receptor / GHS-R1a (via ipamorelin), with research interest in pulsatile growth hormone release, IGF-1 dynamics, body composition endpoints, and receptor selectivity profiles in somatotropic axis investigations.
Benefits
• Dual-pathway activation — combined GHRH receptor + GHS-R agonism studied for synergistic GH release
• DPP-IV resistance — CJC-1295 (no DAC) modifications confer enzymatic stability and extended action vs native GRF
• Ipamorelin selectivity — explored for minimal cortisol, prolactin, and ACTH off-target effects
• Pulsatile preservation — investigated for maintaining physiological GH pulsatility unlike exogenous GH
• IGF-1 signaling — examined for downstream hepatic IGF-1 production and somatotropic endpoints
• Body composition research — investigated in aged rodent models and somatopause endpoints
Localized Effects with Nasal Spray
• Rapid absorption through the nasal mucosa
• Bypasses first-pass hepatic metabolism that limits oral peptide bioavailability
• Convenient delivery route for paired-peptide research protocols requiring repeated dosing
• Co-administration in a single vehicle enables synchronized pharmacokinetic studies
• Formulated with a specialized isotonic nasal vehicle to minimize irritation and dryness
What Researchers Use It For
• Combined GHRH receptor and GHS-R1a pharmacology studies
• Pulsatile GH release dynamics under dual-pathway stimulation
• IGF-1, IGFBP-3, and somatotropic axis endpoint research
• DPP-IV resistance and peptide pharmacokinetic profiling
• Cortisol, prolactin, and ACTH selectivity profiling
• Comparative pharmacology versus other GHRH/GHS-R combinations
Identity & Specs
• Compound 1: CJC-1295 without DAC (Mod GRF 1-29)
• Class: Modified GHRH(1-29) analog with D-Ala², Gln⁸, Ala¹⁵, Leu²⁷ substitutions
• Sequence: Tyr-D-Ala-Asp-Ala-Ile-Phe-Thr-Gln-Ser-Tyr-Arg-Lys-Val-Leu-Ala-Gln-Leu-Ser-Ala-Arg-Lys-Leu-Leu-Gln-Asp-Ile-Leu-Ser-Arg-NH₂
• Formula / M.W.: C₁₅₂H₂₅₂N₄₄O₄₂, ~3367.95 g/mol
• CAS: 863288-34-0
• Compound 2: Ipamorelin
• Class: Selective ghrelin / GHS-R1a agonist (pentapeptide)
• Sequence: Aib-His-D-2-Nal-D-Phe-Lys-NH₂
• Formula / M.W.: C₃₈H₄₉N₉O₅, ~711.85 g/mol
• CAS: 170851-70-4
• Form: Sterile nasal spray solution
• Net per vial: 10 mL (~100 sprays)
• Total Content: 10 mg combined peptide per vial
• Concentration: CJC-1295 (no DAC) 0.5 mg/mL + Ipamorelin 0.5 mg/mL
• Purity: ≥99% by HPLC (per component)
• Identity: MS-verified (per COA)
• Storage: Refrigerate 2–8 °C, protect from light
⚠️ Disclaimer
- This product is intended for laboratory research use only.
- Not for human or veterinary use.
- Not approved for diagnostic, therapeutic, or medical applications.
- Handle using appropriate laboratory safety procedures and personal protective equipment.
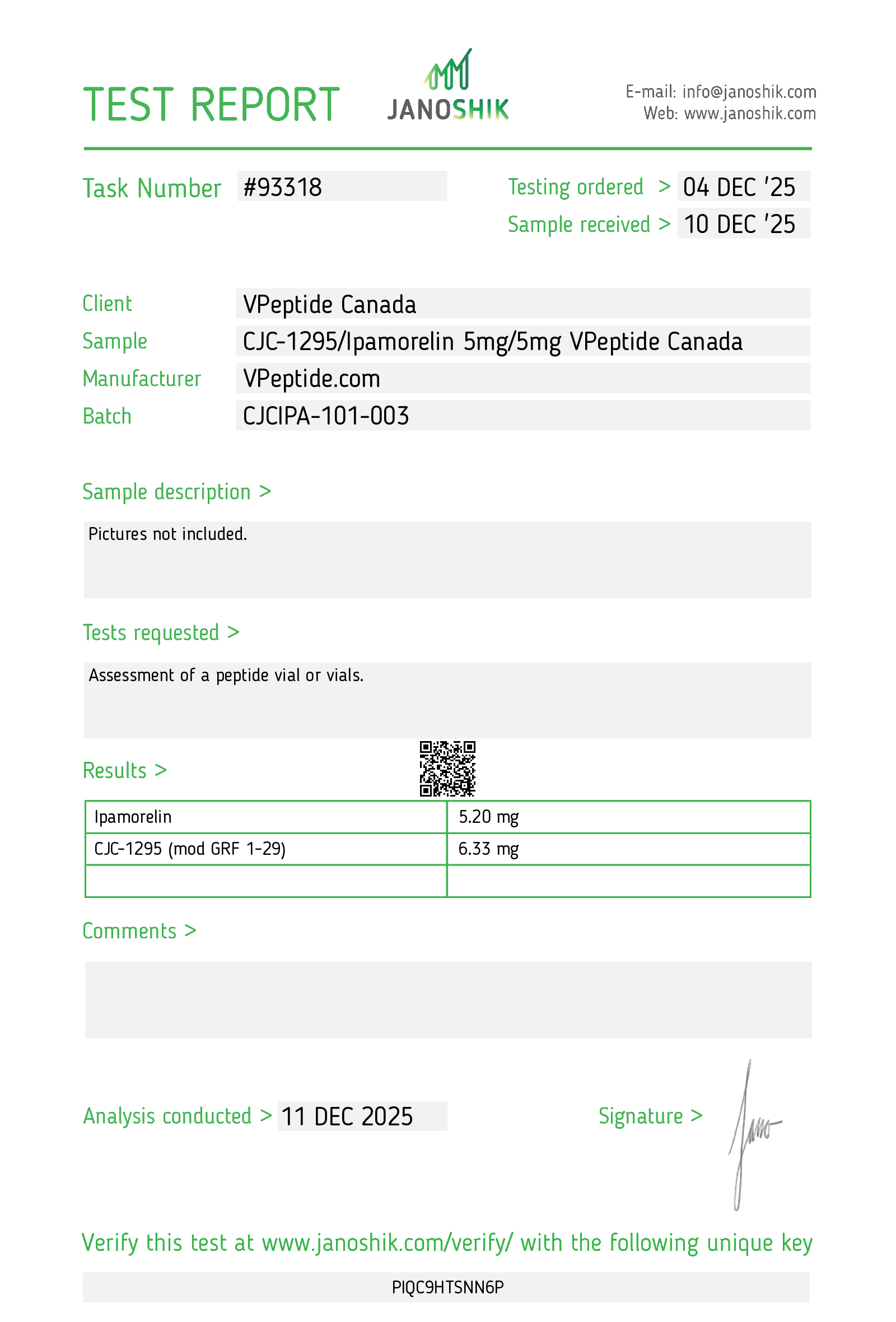
JANOSHIK COA – THIRD-PARTY TESTING
https://janoshik.com/tests/93318-CJC1295Ipamorelin_5mg5mg_VPeptide_Canada_PIQC9HTSNN6P – December 11 2025
121732-CJCIPA_VPeptide_Canada_1UYTSEGTB142 – March 16 2026